Author: Sharon Jang | Reviewer: Tova Plashkes | Published: 24 September 2020 | Updated: 7 December 2021
This page provides an overview of how spinal cord injury (SCI) affects breathing and coughing, and the acute treatments used to address these issues.
Key Points
- Spinal cord injury can damage the muscles of breathing, affecting the ability to take a deep breath, cough and clear mucus, and maintain adequate oxygen levels. The extent of these changes depends on the level and completeness of the SCI with higher cervical injuries being more affected.
- A wide range of management options may be used to assist or improve the effectiveness of breathing and coughing in acute and chronic SCI, including tracheostomy and intubation in severe cases, non-invasive ventilation, along with assisted coughing techniques.
- A number of secondary respiratory complications can affect people with SCI long after injury, including lung infections like pneumonia.
- Preventative strategies such as flu shots, smoking cessation, and healthy living are an important component of respiratory care.

The breathing process: During inhalation, the diaphragm (dark pink) moves down and the ribs expand. During exhalation, the diaphragm moves up and the ribs contract.1
The respiratory system is responsible for helping you breathe in (inhaling) and out (exhaling). Breathing is done through the nose and mouth, although the nose is more often used. One reason the nose is used more often is because it acts as a filter for debris, which protects the lungs. Coughing is another important part of the respiratory system, as it helps to clear mucus from your lungs and airways.
Many muscles help with respiratory functions. The muscles used for inhaling are controlled from the spinal nerves of the neck (C3-C5 primarily), with some help from the nerves of the lower neck and thorax (C6-T12). Inhaling is mostly facilitated by your diaphragm, which is a large dome shaped muscle underneath the lungs. When you breathe in, your diaphragm lowers and the space in your chest increases, pulling air into the lungs. When you exhale, your ribs move back in and the diaphragm moves back up.
Breathing during exercise and coughing requires extra effort. To help, the abdominal muscles (over your belly) and the intercostal muscles (which help to squeeze the ribs) are activated, allowing for a stronger inhalation and a forceful cough.
When someone is unable to breathe or cough by themselves, their function, independence, and health are affected. These respiratory complications arise in 36-83% of individuals after SCI. This is due to disruption of the breathing nerves after an injury, and to secondary complications of an SCI, such as spasticity. After an SCI, breathing muscles may be partially or completely affected, depending on the completeness of the injury. However, breathing ability may improve over time.
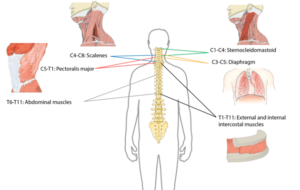
Many muscles are required for breathing. The diagram above shows the main muscles of breathing, and the sections of the spinal cord that innervate them.2
The chances of experiencing respiratory complications depend on a variety of factors, including:
- The level of injury (dictates which muscles are spared)
- The completeness of an injury
- Timing of a tracheostomy*
- The cause of injury
- Age*
* Conflicting evidence
Refer to our page on Evidence Rating for more information on conflicting evidence.
Changes in breathing
Changes in amount of air getting into the lungs
 After injury, the amount of air that can be inhaled and exhaled are significantly reduced for people with cervical and higher thoracic (neck and upper back) level injuries. More specifically, the amount of air that can fill the lungs (known as the total lung capacity) is reduced to 60-80% of normal values. Additionally, the amount of air that can be exhaled after the biggest breath in (known as the vital capacity) is reduced to 50-80% of normal values. This contributes to inefficient breathing that may be tiresome and difficult. In addition, a lower vital capacity can impact voice volume, making it difficult to speak at louder volumes.
After injury, the amount of air that can be inhaled and exhaled are significantly reduced for people with cervical and higher thoracic (neck and upper back) level injuries. More specifically, the amount of air that can fill the lungs (known as the total lung capacity) is reduced to 60-80% of normal values. Additionally, the amount of air that can be exhaled after the biggest breath in (known as the vital capacity) is reduced to 50-80% of normal values. This contributes to inefficient breathing that may be tiresome and difficult. In addition, a lower vital capacity can impact voice volume, making it difficult to speak at louder volumes.
Changes to the lung
An SCI can affect the lung itself. The main change is a reduction in lung compliance, or the lung’s ability to stretch and expand. As a result, the lung does not spring back “closed” after being open. In addition, the compliance of the rib cage (chest wall) may also decrease, causing the chest to become rigid in individuals with tetraplegia. Reduced compliance results in a decreased ability to take a deep breath independently or with the help of a breathing bag or ventilator.
Changes in coughing
![]() Coughing is important to keep the airway and lungs clear from mucus. This is because a build up of mucus can collapse the lungs, and mucus in the airways can result in infection. In order to perform a cough, one needs to inhale deeply then have a forceful exhale while a structure called the glottis closes the entrance to the windpipe. The intercostal muscles and the abdominal muscles assist with the ability to increase the force of exhaled air. As these muscles are innervated by nerves in the chest region, individuals with spinal cord injuries may have an impaired coughing function. Cough function may be completely absent in some individuals, while others may have limited or ineffective coughing abilities.
Coughing is important to keep the airway and lungs clear from mucus. This is because a build up of mucus can collapse the lungs, and mucus in the airways can result in infection. In order to perform a cough, one needs to inhale deeply then have a forceful exhale while a structure called the glottis closes the entrance to the windpipe. The intercostal muscles and the abdominal muscles assist with the ability to increase the force of exhaled air. As these muscles are innervated by nerves in the chest region, individuals with spinal cord injuries may have an impaired coughing function. Cough function may be completely absent in some individuals, while others may have limited or ineffective coughing abilities.
Changes in lung irritability and mucous production
Soon after injury, it is common for individuals with high-level SCI to produce a lot of mucus in their lungs and have smaller airways deep in the lung. The lungs are also very irritable to stimuli like too much suctioning of mucus, or smoking. This may be due to the increased influence of the parasympathetic nervous system after SCI. In people with acute tetraplegia, it has been reported that an excess of up to 1 liter of mucus is produced each day. In combination with an inability to cough, this excess production of mucus can result in a buildup of fluid in the lungs and airway.
Changes in swallowing
Although swallowing is important for eating, it is also important for clearing the throat to prevent food, drink, stomach contents, or saliva from entering the lungs (also known as aspiration). After SCI, the risk of aspiration increases as:
- Your ability to cough may be limited by medical conditions and weakness due to your injury
- Surgical procedures on the spine may compress your throat
- You may feel less alert due to sedative medications
- Some medications you may be on can lead to dry mouth
- Your sensation may be impaired, which prevents you from feeling food or liquid in the spaces at the back of your throat
The lack of effective swallowing can cause mucus to collect in your airway. Over time, the stagnant mucus can encourage the growth of bacteria, which may travel down to your lungs and potentially result in pneumonia.
Secretion Removal Techniques
Efficient removal of mucus from the airways is important to prevent choking and lung infections, especially when independent coughing is difficult. Although research on the topic of secretion removal techniques is scarce, one study with moderate evidence showed that manual removal techniques combined with mechanical removal techniques are effective in SCI early after injury. Different techniques are outlined below:
Postural drainage
Certain body positions can use gravity to drain mucus towards the throat to be excreted easier. For example, laying on your side with your feet elevated can help drain the lower lung. In order for these positions to be effective, your body must be positioned in specific angles. Refer to your healthcare professional for more information. To facilitate breakup and movement of the mucus buildup in the lungs, postural drainage can be paired with applying pressure to the chest (chest percussion) or shaking the chest (vibration).
Manual assisted coughing
Physical pressure is applied to the chest or abdomen right before expiration to help the individual breathe out. This can be done on yourself or by a trained family member or caregiver.
Mechanically assisted coughing (insufflation-exsufflation)
There are machines that help loosen secretions, clear mucus, and can trigger a cough. They work by delivering a deep breath by pushing air into the lungs, then facilitate exhalation by sucking the air out.
Suction
A tube can be inserted through the mouth or tracheostomy site to suction mucus that is stuck in the upper airways. Suctioning may also reflexively trigger a cough.
Respiratory Muscle Training
Weak inspiratory muscles can result in breathlessness. Like exercise training, inspiratory muscle strength and endurance can increase with training and decrease bouts of shortness of breath (dyspnea) and coughing. Inspiratory muscle training involves using devices that create resistance when breathing in.

Resistive trainers have adjustable settings that allow individualized training programs.5
Refer to our article on Inspiratory Muscle Training for more information!
Drug Treatments
Bronchodilators
People with tetraplegia have increased sensitivity of their airways, resulting in more frequent narrowing. To treat this, a family of drugs called bronchodilators can be used to enlarge the airways for air to pass through with more ease. The use of bronchodilators is supported by multiple (weak evidence) studies, which have found that bronchodilators can help improve expiration among individuals with tetraplegia. There is also one strong evidence study that indicates that the bronchodilator salmeterol can improve both respiratory functioning and the strength of breathing muscles. While bronchodilators can help positively influence respiratory functioning, their use carries a potential negative side effect of thickening mucus.
Mechanical ventilation, or machine assisted breathing, is becoming more common as there has been an increase in the number of people who survive cervical level injuries over the past 40 years. Mechanical ventilation is used by people who are unable to breathe independently, often right after injury. The machine works by pushing air into the lungs until a pre-set volume or pressure is reached. Once the pre-set value has been met, the machine stops pushing air in and the air is exhaled by the person.
In general, there are two forms of mechanical ventilation: a non-invasive approach where a mask is placed over the mouth and nose (known as a Bilevel Positive Airway Pressure (BiPAP) or Continuous Positive Airway Pressure (or CPAP)), or an invasive approach where a tube is inserted into the windpipe via the mouth and throat (intubation) or directly into the windpipe through a surgical incision (tracheostomy). Intubation or tracheostomy is used in more severe cases to ensure air gets to the lungs and that mucus is filtered out of the lung. Factors that increase the chances of requiring invasive mechanical ventilation include having a complete injury, having a higher level of injury, or having a compound injury Whenever possible, the healthcare team tries to help people breathe on their own and “weans” the person off the ventilator if possible.
Intubation
The process of intubation consists of running a tube into the trachea either through the nose or mouth. This process is completed as soon as someone is in respiratory distress, which is normally at the scene of the accident or upon admission to the hospital. Intubation is often used for a short term (i.e., less than 10 days), as prolonged use can lead to severe weakness of breathing muscles, pneumonia, more difficulty in breathing, mobility limitations, prolonged ventilator weaning, and can make lung and mouth hygiene difficult.
Tracheostomy
A tracheostomy is a surgical procedure that involves placing a tube through an opening in the throat and windpipe. This creates a pathway for air delivery from a ventilator and to facilitate secretion removal. However, after the tracheostomy tube is taken out, speaking and eating may be difficult as the throat muscles become weakened and uncoordinated.
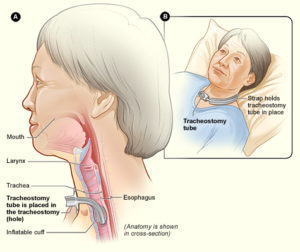
A tracheostomy tube is inserted into the throat through a surgical hole.6
A tracheostomy is performed in the event that breathing support is required for a minimum of 3 weeks. Individuals who may require a tracheostomy (weak to moderate evidence) include: having a complete or higher level of injury, having a complete injury or a lower AIS motor grade, and old age.
Once an individual is able to independently breathe, the tube is removed from the windpipe. Weak evidence suggests that tube removal is more successful in individuals with certain characteristics:
- Those who have lower level spinal cord injuries.
- Those who have not had a tracheostomy but have only been intubated.
Continuous Positive Airway Pressure (CPAP) Ventilation
Continuous Positive Airway Pressure (CPAP) is a form of mechanical ventilation commonly used to address obstructive sleep apnea. Sleep apnea occurs when breathing ceases in short bouts during sleep, and can result in feeling tired during the day. CPAP machines are used to manage this condition by acting as a “pneumatic splint”, keeping airways open during sleep.
Loss of independent breathing and cough function can lead to secondary respiratory issues. These issues need to be medically addressed, as they may be life-threatening if left untreated.
Common secondary respiratory issues following SCI
Atelectasis
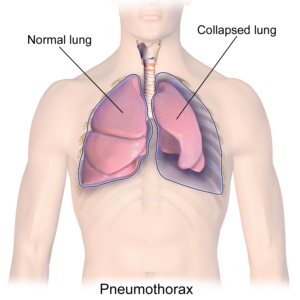
A collapsed lung in comparison to a healthy lung.7
A condition where a part of the lung becomes partially or fully collapsed due to a lack of air. This results in a reduced ability to exchange oxygen and carbon dioxide. When the body does not get enough oxygen, organs will start to shut down. Atelectasis can result from anything that prevents the lungs from fully expanding, including:
- Weak or paralyzed muscles, which can prevent being able to take in a deep full breath. This is the most important cause in SCI.
- A buildup of mucus, which may block an area of the lung from fully expanding.
- Shallow breathing due to surgery or pain, which can result in poor inflation of the lungs.
Pneumonia
Pressure from outside the lungs, which can result in the inability to fully inflate. This external pressure may stem from fluid or air, abdominal organs, or external hardware such as a brace.
Pneumonia is a medical name for a lung infection. After an SCI, several factors make pneumonia very common:
Requiring a ventilator, suctioning (removal of secretions with a special machine), or a tracheostomy may often be necessary, but tends to introduce bacteria despite best efforts at hygiene and air filtration. The risk of getting pneumonia increases if someone:
- is unable to cough and clear mucus,
- is reliant on mechanical ventilation to assist with breathing,
- has a severe injury,
- has a traumatic higher-level injury involving fractures, or,
- has had a surgical tracheostomy.
Refer to our article on Infectious Respiratory Conditions for more information!
Pulmonary embolism
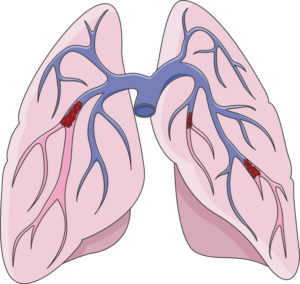
A pulmonary embolism occurs when a clot (red groups) gets caught in the lungs.8
A pulmonary embolism is a blockage of an artery in the lungs by a blood clot that has moved from elsewhere in the body through the bloodstream (embolism). As a result of paralysis or immobility, a blockage may develop in a vein, often in the lower leg. This is called a blood clot, or deep vein thrombosis. The clot may travel to the lung and block blood vessels, resulting in sudden shortness of breath. The prevalence of this condition is highest within the first three months of injury. Weak evidence suggests that pulmonary embolisms occur in a range of 1.25% to 4.5% of people with SCI in the first 90 days. However, pulmonary embolisms have been considered to occur rarely after the first three months of SCI, and have decreased significantly due to preventative measures (e.g., blood thinners). That said, weak evidence suggests that pulmonary embolisms may still be an issue in chronic SCI, but may not be severe enough to cause any symptoms.
Pulmonary edema
Pulmonary edema is a build-up of fluid in the lung. This often occurs in early stages following injury. It can affect as much as 50% of individuals with acute tetraplegia. There are several causes, with the most common being excess fluids given to people with SCI. After an SCI, blood pressure may drop to very low levels. Depending on the cause and type of injury, this may be due to blood loss from a traumatic injury, or impairment of nerves that keep blood pressure at its normal level with a cervical or high thoracic injury. As a result, a lot of fluids are given to patients to help their blood pressure recover.
Respiratory failure
Respiratory failure occurs when the respiratory system is damaged to the extent where the body does not get enough oxygen and is unable to get rid of carbon dioxide. Oxygen levels in the body may drop to critically low values and carbon dioxide, which is poisonous at very high levels, builds up. The risk of a respiratory failure increases with higher levels of injury, and most commonly occurs in acute SCI. This usually results in the need for mechanical ventilation.
Getting vaccinated is one of the ways to prevent secondary complications such as pneumonia.9
Prevention is important to avoid getting respiratory illnesses when you have an SCI. Some things you can do to stay as healthy as possible include:
- Avoiding smoking any substances and taking in second hand smoke. The lungs of people with SCI are easily irritated, and those who smoke are more susceptible to lung infections.
- Staying hydrated – drink plenty of water. This helps to keep mucus in the lungs from being too thick.
- Ensuring proper nutrition to help maintain a healthy weight and ensure the body has enough vitamins, minerals and protein to heal well when sick.
- Exercising, as it can help by:
- Helping you maintain a healthy weight, as lung complications become more prevalent in people who are overweight or obese,
- Strengthening your breathing muscles.
- Getting vaccinated for influenza (the flu) and pneumonia. This can help decrease your odds of getting these illnesses.
- Coughing on a regular basis. Coughing is important for keeping your airways clear of secretions. If you have difficulties coughing by yourself, have someone help you perform manual assist coughs, or use a cough assist machine.
- Maintaining mobility and proper posture. In order to prevent build up in the lungs, try to sit up everyday and turn when laying in bed.
Secretion removal techniques
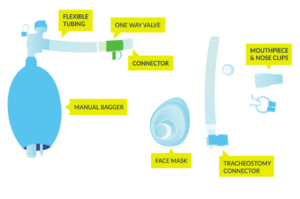
Equipment used for lung volume recruitment.10
Upon returning to the community, common secretion removal techniques include glossopharyngeal breathing and the use of lung volume recruitment (LVR) bags with an assisted cough. Glossopharyngeal breathing (or frog breathing) is a technique that is used to get a deeper breath. This is done by rapidly taking “gulps” of breaths one after the other, followed by exhaling. This can help create a cough, or facilitate assisted coughing.
LVR, or “breath stacking” is done with an LVR kit which consists of a resuscitation bag connected with a flexible tube to a mouth piece with a one-way valve. The individual will inhale the most they can, and once this point is reached, a clinician (or second person) will squeeze the bag to “stack” breaths to fully inflate the lungs. This allows the individual to breathe more air than they are able to themselves, and to exhale more air more quickly to produce an improved cough. This also can help with maintaining chest mobility and flexibility.
Exercise Training
 Exercise training involving arm and leg movements can improve muscle strength and cardiovascular endurance. Breathing muscles are also challenged with exercise and may become stronger with exercise. This increase in strength can help decrease the effort of breathing at rest and with functional activity, like transfers. An example of a method of exercise training for individuals with higher levels of injuries include the use of a Functional Electrical Stimulation (FES) bike. Other exercises like arm cycling or strengthening exercises are commonly prescribed by a physiotherapist or health care professional. While exercise can help strengthen respiratory muscles, low-moderate evidence studies debate whether lung volumes are impacted. This is to say, exercising may help make breathing feel easier, but it is unknown whether the amount of air you can take into your lungs is affected. High intensity exercise three times per week for six weeks has shown to significantly improve respiratory function. However, standard guidelines for high intensity exercise have not yet been established.
Exercise training involving arm and leg movements can improve muscle strength and cardiovascular endurance. Breathing muscles are also challenged with exercise and may become stronger with exercise. This increase in strength can help decrease the effort of breathing at rest and with functional activity, like transfers. An example of a method of exercise training for individuals with higher levels of injuries include the use of a Functional Electrical Stimulation (FES) bike. Other exercises like arm cycling or strengthening exercises are commonly prescribed by a physiotherapist or health care professional. While exercise can help strengthen respiratory muscles, low-moderate evidence studies debate whether lung volumes are impacted. This is to say, exercising may help make breathing feel easier, but it is unknown whether the amount of air you can take into your lungs is affected. High intensity exercise three times per week for six weeks has shown to significantly improve respiratory function. However, standard guidelines for high intensity exercise have not yet been established.
An abdominal binder wrapped around the abdomen to correct the positioning of the diaphragm.12
Refer to our article on Functional Electrical Stimulation for more information!
Girdle/Abdominal Binder
Girdles or abdominal binders are garments that apply pressure around the abdominal area to help keep the diaphragm in an optimal position. Abdominal binders are also used for managing orthostatic hypotension and blood pooling. Although there may be short-term improvements when using a girdle or binder, more research is needed in determining their long-term utility.
Refer to our article on Abdominal binders for more information!
Electrical Stimulation
For people who are ventilator dependent, various electrical stimulation techniques are available to assist with breathing. This includes phrenic nerve stimulation/diaphragm pacing, abdominal electrical stimulation, and epidural stimulation.
Phrenic nerve stimulation/Diaphragm pacing
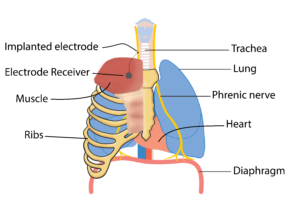 The diaphragm is the main muscle responsible for breathing and is activated by the phrenic nerve to contract. After SCI, the connection between the brain and the phrenic nerve is disrupted, which contributes to impaired breathing. Researchers have been looking at ways we can stimulate this nerve to reactivate the diaphragm through a process called phrenic nerve stimulation. This process involves surgically implanting electrodes and a receiver close to the phrenic nerve, either in the chest or the neck, and a receiver in the chest wall. This device is controlled with an external remote and antenna (which is used to connect to the electrode receiver).
The diaphragm is the main muscle responsible for breathing and is activated by the phrenic nerve to contract. After SCI, the connection between the brain and the phrenic nerve is disrupted, which contributes to impaired breathing. Researchers have been looking at ways we can stimulate this nerve to reactivate the diaphragm through a process called phrenic nerve stimulation. This process involves surgically implanting electrodes and a receiver close to the phrenic nerve, either in the chest or the neck, and a receiver in the chest wall. This device is controlled with an external remote and antenna (which is used to connect to the electrode receiver).
For phrenic nerve stimulation to work, the diaphragm must have normal function, and the phrenic nerve needs to be intact (i.e., sends a signal when stimulated). As a result, individuals who have a C3, C4, or C5 level injury may not be eligible as they often have impaired phrenic nerve function. It is important to note that this procedure can only facilitate inspiratory functions, but not expiratory. As a result, an individual who receives phrenic nerve stimulation may not require mechanical ventilation, but will still require assistance with coughing and clearing secretions. Tracheostomies and mechanical ventilation are often still used in combination with phrenic nerve stimulation as a back-up.
Some weak evidence supports the use of phrenic nerve stimulation. One study found that diaphragm pacemakers have better results with long term implantation (i.e., 6.3 years in the study). Another study showed that diaphragmatic paces can improve survival rates, decrease the cost of care, improve the quality of speech, increase rates of social participation, and improve management of using a powered wheelchair. Many complications have been reported in the research in regards to using a phrenic nerve stimulator. These complications include wires breaking or getting displaced, device failure, inhaling food when eating, shoulder or abdominal pain, and infections.
Abdominal electrical stimulation
As diaphragm pacing only helps with inhalation, limited research suggests that electrically stimulating the abdominal muscles helps with expiration and coughing. Ideally, the abdominal muscles would be used to support voluntary efforts to cough. There have been mixed findings on the effectiveness of stimulating the abdominal muscles to enhance cough. While some weak studies have found abdominal stimulation to improve cough, other weak evidence studies have found no noticeable changes. More research is required to determine the efficacy of stimulating the abdominal muscles to enhance cough in SCI.
Epidural Stimulation
Epidural stimulation is conducted through surgically implanting an electrode over the spinal cord. Once done, the electrode, which is controlled with a remote outside of the body, stimulates various parts of the spinal cord. Emerging research on epidural stimulation suggests that it may benefit respiratory function after SCI. By directly stimulating nerve cells in the spinal cord, weak evidence suggests that breathing muscles can be activated. The muscles are activated in a pattern that resembles normal breathing, while reducing fatigue. Additionally, weak evidence suggests that epidural stimulation can improve other respiratory functions including coughing and speaking.
Respiratory problems are common after SCI. The extent and type of these problems depend on the level of injury and completeness of injury. Both conservative and invasive options for managing respiratory health following an SCI are available. Due to impaired respiratory function, a variety of secondary complications to the lungs frequently occur after SCI. While prevention using proper respiratory hygiene is best, should you experience a secondary respiratory complication, a variety of management techniques can be applied. Some techniques are more common in the acute stages of SCI, while others are more suited to chronic SCI. It is best to discuss all treatment options with your health providers to find out which treatments are suitable for you.
Mullen E, Mirkowski M, Vu V, McIntyre A, Teasell RW. (2015). Respiratory Management during the Acute Phase of Spinal Cord Injury. In Eng JJ, Teasell RW, Miller WC, Wolfe DL, Townson AF, Hsieh JTC, Connolly SJ, Noonan VK, Loh E, McIntyre A, editors. Spinal Cord Injury Research Evidence. Version 5.0: p 1-50.
Available from: https://scireproject.com/evidence/acute-evidence/respiratory-management-during-acute-phase-of-spinal-cord-injury/
Evidence for “How does an SCI affect the respiratory system” is based on
Warren, P. M., Awad, B. I., & Alilain, W. J. (2014). Drawing breath without the command of effectors: The control of respiration following spinal cord injury. Respiratory Physiology & Neurobiology. https://doi.org/10.1016/j.resp.2014.08.005
Lemons, V. R., & Wagner, F. C. (1994). Respiratory Complications After CSCI. In Spine (Vol. 19, Issue 20, pp. 2315–2320).
Romero-Ganuza, J., Gambarrutta, C., Merlo-Gonzalez, V. E., Marin-Ruiz, M. Á., Diez De La Lastra-Buigues, E., & Oliviero, A. (2011). Complications of tracheostomy after anterior cervical spine fixation surgery. American Journal of Otolaryngology – Head and Neck Medicine and Surgery, 32(5), 408–411. https://doi.org/10.1016/j.amjoto.2010.07.020
Romero, J., Vari, A., Gambarrutta, C., & Oliviero, A. (2009). Tracheostomy timing in traumatic spinal cord injury. European Spine Journal, 18(10), 1452–1457. https://doi.org/10.1007/s00586-009-1097-3
Aarabi, B., Harrop, J. S., Tator, C. H., Alexander, M., Dettori, J. R., Grossman, R. G., Fehlings, M. G., Mirvis, S. E., Shanmuganathan, K., Zacherl, K. M., Burau, K. D., Frankowski, R. F., Toups, E., Shaffrey, C. I., Guest, J. D., Harkema, S. J., Habashi, N. M., Andrews, P., Johnson, M. M., & Rosner, M. (2012). Predictors of pulmonary complications in blunt traumatic spinal cord injury. Journal of Neurosurgery: Spine, 17, 38–45.
Jain, N. B., Higgins, L. D., Katz, J. N., & Garshick, E. (2010). Association of shoulder pain with the use of mobility devices in persons with chronic spinal cord injury. PM and R, 2(10), 896–900. https://doi.org/10.1016/j.pmrj.2010.05.004
Anke, A., Aksnes, A. K., Stanghelle, J. K., & Hjeltnes, N. (1993). Lung volumes in tetraplegic patients according to cervical spinal cord injury level. Scandinavian Journal of Rehabilitation Medicine, 25(2), 73–77. http://www.ncbi.nlm.nih.gov/pubmed/8341994
Brown, R., DiMarco, A. F., Hoit, J. D., & Garshick, E. (2006). Respiratory dysfunction and management in spinal cord injury. Respiratory Care, 51(8), 853-68;discussion 869-70. http://www.ncbi.nlm.nih.gov/pubmed/16867197%0Ahttp://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=PMC2495152
Berlly, M., & Shem, K. (2007). Respiratory management during the first five days after spinal cord injury. Journal of Spinal Cord Medicine, 30(4), 309–318. https://doi.org/10.1080/10790268.2007.11753946
Schilero, G. J., Spungen, A. M., Bauman, W. A., Radulovic, M., & Lesser, M. (2009). Pulmonary function and spinal cord injury. Respiratory Physiology and Neurobiology, 166(3), 129–141. https://doi.org/10.1016/j.resp.2009.04.002
Schilero, G.J., Grimm, D.R., Bauman, W.A., Lenner, R., Lesser, M. (2005). Assessment of airway caliber and bronchodilator responsiveness in subjects with spinal cord injury. Chest, 127(1), 149-155. http://dx.doi.org/10.1378/chest.127.1.149
Bhaskar, K. R., Brown, R., O’Sullivan, D. D., Melia, S., Duggan, M., & Reid, L. (1991). Bronchial Mucus Hypersecretion in Acute Quadriplegia: Macromolecular Yields and Glycoconjugate Composition. American Review of Respiratory Disease, 143(3), 640–648. https://doi.org/10.1164/ajrccm/143.3.640
Chaw, E., Shem, K., Castillo, K., Wong, S., & Chang, J. (2012). Dysphagia and associated respiratory considerations in cervical spinal cord injury. Topics in Spinal Cord Injury Rehabilitation, 18(4), 291–299. https://doi.org/10.1310/sci1804-291
Evidence for “How does an SCI affect the respiratory system” is based on
Almenoff PL, Alexander LR, Spungen AM, Lesser MD, Bauman WA. Bronchodilatory effects of ipratropium bromide in patients with tetraplegia. Paraplegia 1995; 33: 274-7.
Spungen AM, Dicpinigaitis PV, Almenoff PL, Bauman WA. Pulmonary obstruction in individuals with cervical spinal cord lesions unmasked by bronchodilator administration. Paraplegia 1993;31:404-7.
Schilero GJ, Grimm D, Spungen AM, Lenner R, Lesser M. Bronchodilator responses to metaproterenol sulfate among subjects with spinal cord injury. J Rehabil Res Dev 2004; 41: 59-64.
Grimm DR, Schilero GJ, Spungen AM, Bauman WA, Lesser M. Salmeterol improves pulmonary function in persons with tetraplegia. Lung 2006; 184: 335–339.
Evidence for “How can mechanical ventilation be used to help with breathing” is based on
Devivo, M. J. (2012). Epidemiology of traumatic spinal cord injury: Trends and future implications. Spinal Cord, 50(5), 365–372. https://doi.org/10.1038/sc.2011.178
Gregoretti, C., Olivieri, C., & Navalesi, P. (2005). Physiologic comparison between conventional mechanical ventilation and transtracheal open ventilation in acute traumatic quadriplegic patients. Critical Care Medicine, 33(5), 1114–1118. https://doi.org/10.1097/01.CCM.0000162559.74446.09
Como, J. J., Sutton, E. R. H., McCunn, M., Dutton, R. P., Johnson, S. B., Aarabi, B., & Scalea, T. M. (2005). Characterizing the need for mechanical ventilation following cervical spinal cord injury with neurologic deficit. Journal of Trauma – Injury, Infection and Critical Care, 59(4), 912–916. https://doi.org/10.1097/01.ta.0000187660.03742.a6
Montoto-Marqués, A., Trillo-Dono, N., Ferreiro-Velasco, M. E., Salvador-De La Barrera, S., Rodriguez-Sotillo, A., Mourelo-Fariña, M., Galeiras-Vázquez, R., & Meijide-Failde, R. (2018). Risks factors of mechanical ventilation in acute traumatic cervical spinal cord injured patients. Spinal Cord, 56(3), 206–211. https://doi.org/10.1038/s41393-017-0005-7
Seidl, R. O., Wolf, D., Nusser-Müller-Busch, R., & Niedeggen, A. (2010). Airway management in acute tetraplegics: A retrospective study. European Spine Journal, 19(7), 1073–1078. https://doi.org/10.1007/s00586-010-1328-7
Velmahos, G. C., Toutouzas, K., Chan, L., Tillou, A., Rhee, P., Murray, J., & Demetriades, D. (2003). Intubation after cervical spinal cord injury: to be done selectively or routinely? The American Surgeon, 69, 891–894. https://doi.org/10.1257/0002828041464551
Durbin, C. G., Bell, C. T., & Shilling, A. M. (2014). Elective intubation. Respiratory Care, 59(6), 825–849.
Shirawi, N., & Arabi, Y. (2005). Bench-to-bedside review: Early tracheostomy in critically ill trauma patients. In Critical Care (Vol. 10, Issue 1). BioMed Central Ltd. https://doi.org/10.1186/cc3828
Biering-Sorensen, M. (1992). Tracheostomy in spinal cord injured: frequency and follow up. 30, 656–660.
Ganuza, J. R., Forcada, A. G., Gambarrutta, C., Buigues, E. D. D. L. L., Gonzalez, V. E. M., Fuentes, F. P., & Luciani, A. A. (2011). Effect of technique and timing of tracheostomy in patients with acute traumatic spinal cord injury undergoing mechanical ventilation. Journal of Spinal Cord Medicine, 34(1), 76–84. https://doi.org/10.1179/107902610X12886261091875
McCully, B. H., Fabricant, L., Geraci, T., Greenbaum, A., Schreiber, M. A., & Gordy, S. D. (2014). Complete cervical spinal cord injury above C6 predicts the need for tracheostomy. American Journal of Surgery, 207(5), 668–669. https://doi.org/10.1016/j.amjsurg.2014.01.001
Yugué, I., Okada, S., Ueta, T., Maeda, T., Mori, E., Kawano, O., Takao, T., Sakai, H., Masuda, M., Hayashi, T., Morishita, Y., & Shiba, K. (2012). Analysis of the risk factors for tracheostomy in traumatic cervical spinal cord injury. Spine, 37(26), 1633–1638. https://doi.org/10.1097/BRS.0b013e31827417f1
Leelapattana, P., Fleming, J. C., Gurr, K. R., Bailey, S. I., Parry, N., & Bailey, C. S. (2012). Predicting the need for tracheostomy in patients with cervical spinal cord injury. Journal of Trauma and Acute Care Surgery, 73(4), 880–884. https://doi.org/10.1097/TA.0b013e318251fb34
Menaker, J., Kufera, J. A., Glaser, J., Stein, D. M., & Scalea, T. M. (2013). Admission ASIA motor score predicting the need for tracheostomy after cervical spinal cord injury. Journal of Trauma and Acute Care Surgery, 75(4), 629–634. https://doi.org/10.1097/TA.0b013e3182a12b86
O’Keeffe, T., Goldman, R. K., Mayberry, J. C., Rehm, C. G., & Hart, R. A. (2004). Tracheostomy after anterior cervical spine fixation. Journal of Trauma – Injury, Infection and Critical Care, 57(4), 855–860. https://doi.org/10.1097/01.TA.0000083006.48501.B2
Harrop, J. S., Sharan, A. D., Scheid, E. H., Vaccaro, A. R., & Przybylski, G. J. (2004). Tracheostomy placement in patients with complete cervical spinal cord injuries: American Spinal Injury Association Grade A. Journal of Neurosurgery, 100(Spine 1), 20–23.
Peterson, W., Charlifue, W., Gerhart, A., & Whiteneck, G. (1994). Two methods of weaning persons with quadriplegia from mechanical ventilators. Paraplegia, 32(2), 98–103. https://doi.org/10.1038/sc.1994.17
Kornblith, L. Z., Kutcher, M. E., Callcut, R. A., Redick, B. J., Hu, C. K., Cogbill, T. H., Baker, C. C., Shapiro, M. L., Burlew, C. C., Kaups, K. L., DeMoya, M. A., Haan, J. M., Koontz, C. H., Zolin, S. J., Gordy, S. D., Shatz, D. V, Paul, D. B., Cohen, M. J., & Western Trauma Association Study Group. (2013). Mechanical ventilation weaning and extubation after spinal cord injury: a Western Trauma Association multicenter study. The Journal of Trauma and Acute Care Surgery, 75(6), 1060–1069; discussion 1069-70. https://doi.org/10.1097/TA.0b013e3182a74a5b
Evidence for “What secondary respiratory issues occur with acute SCI?” is based on
Berlly, M., & Shem, K. (2007). Respiratory management during the first five days after spinal cord injury. Journal of Spinal Cord Medicine, 30(4), 309–318. https://doi.org/10.1080/10790268.2007.11753946
Garcia-Arguello, L. Y., O’Horo, J. C., Farrell, A., Blakney, R., Sohail, M. R., Evans, C. T., & Safdar, N. (2017). Infections in the spinal cord-injured population: a systematic review. Spinal Cord, 55(6), 526–534. https://doi.org/https://dx.doi.org/10.1038/sc.2016.173
Alabed, S., De Heredia, L. L., Naidoo, A., Belci, M., Hughes, R. J., & Meagher, T. M. (2015). Incidence of pulmonary embolism after the first 3 months of spinal cord injury. Spinal Cord, 53(11), 835–837. https://doi.org/10.1038/sc.2015.105
Aito, S., Pieri, A., D’Andrea, M., Marcelli, F., & Cominelli, E. (2002). Primary prevention of deep venous thrombosis and pulmonary embolism in acute spinal cord injured patients. Spinal Cord, 40(6), 300–303. https://doi.org/10.1038/sj.sc.3101298
Frisbie, J. H., & Sharma, G. V. R. K. (2012). The prevalence of pulmonary embolism in chronically paralyzed subjects: A review of available evidence. Spinal Cord, 50(6), 400–403. https://doi.org/10.1038/sc.2011.154
Sezer, N., Akkuş, S., & Uğurlu, F. G. (2015). Chronic complications of spinal cord injury. World Journal of Orthopaedics, 6(1), 24–33. https://doi.org/10.5312/wjo.v6.i1.24
Evidence for “What is the emerging research on processes to help with breathing” is based on
DiMarco, A. F., Kowalski, K. E., Hromyak, D. R., & Geertman, R. T. (2014). Long-term follow-up of spinal cord stimulation to restore cough in subjects with spinal cord injury. The Journal of Spinal Cord Medicine, 37(4), 380–388. https://doi.org/10.1179/2045772313Y.0000000152
Carter, R. E. (1993). Experience with ventilator dependent patients ! In Paraplegia (Vol. 31). https://doi.org/10.1038/sc.1993.28
Hirschfeld, S., Exner, G., Luukkaala, T., & Baer, G. A. (2008). Mechanical ventilation or phrenic nerve stimulation for treatment of spinal cord injury-induced respiratory insufficiency. Spinal Cord, 46(11), 738–742. https://doi.org/10.1038/sc.2008.43
Esclarin, A., Bravo, P., Arroyo, O., Mazaira, J., Garrido, H., & Alcaraz, M. A. (1994). Tracheostomy ventilation versus diaphragmatic pacemaker ventilation in high spinal cord injury. Paraplegia, 32(10), 687–693. https://doi.org/10.1038/sc.1994.111
DiMarco, A. F., Kowalski, K. E., Geertman, R. T., & Hromyak, D. R. (2006). Spinal cord stimulation: a new method to produce an effective cough in patients with spinal cord injury. American Journal of Respiratory and Critical Care Medicine, 173(12), 1386–1389. https://doi.org/10.1164/rccm.200601-097CR
Hachmann, J. T., Grahn, P. J., Calvert, J. S., Drubach, D. I., Lee, K. H., & Lavrov, I. A. (2017). Electrical Neuromodulation of the Respiratory System After Spinal Cord Injury. Mayo Clinic Proceedings, 92(9), 1401–1414. https://doi.org/10.1016/j.mayocp.2017.04.011
Harkema, S. J., Wang, S., Angeli, C. A., Chen, Y., Boakye, M., Ugiliweneza, B., & Hirsch, G. A. (2018). Normalization of Blood Pressure With Spinal Cord Epidural Stimulation After Severe Spinal Cord Injury. Frontiers in Human Neuroscience, 12, 83. https://doi.org/10.3389/fnhum.2018.00083
Image Credits:
- The breathing process ©The SCIRE Community Team
- Modified from: Musculi colli base © Olek Remesz, CC-BY-SA 2.5; Muscles that move the humerus ©OpenStax, CC BY 4.0; Thorax ©OpenStax, CC BY 4.0; Respiratory System ©Theresa Knott, CC BY-SA 3.0, Vertebral Column ©Servier Medical Art, CC BY 3.0; Outline ©Servier Medical Art, CC BY 3.0
- Lungs ©Mahmure Alp, CC BY 3.0
- Sneezing icon ©j4p4n, CC 0
- POWERbreathe Plus, ©POWERbreathe
- Tracheostomy NIH ©National Heart Lung and Blood Institute, CC 0
- Blausen 0742 Pneumothorax ©Bruce Blaus, CC BY 3.0
- Pulmonary embolism ©Servier Medical Art, CC BY 3.0
- Cure medical care medication pharmacology vaccination ©Bicanski, CC 0
- Lung volume recruitment set ©The SCIRE Community Team
- Using the FES ©The SCIRE Community Team
- Abdominal Binder ©The SCIRE Community Team
- Modified from: Diagram 1 of 3 showing stage 3A lung cancer CRUK 008 ©Cancer Research UK, CC BY 4.0; Breathing ©Servier Medical Art, CC BY 3.0





